The obtained results under optimal conditions (SMZ concentration = 20 ppm, pH = 6, irradiation time = 120 min) were as follows: removal efficiency of 91.7%, P max = 16.98 μW/cm 2, J sc = 96.75 μA/cm 2, V oc = 644 mV. 
Additionally, polarization and electrochemical tests such as chronoamperometry and EIS were measured to further examine the electrochemical features of the PFC photoanode system. In order to investigate the properties of the photocatalyst, structural analyses including XRD, FESEM, FTIR, and UV–vis DRS were utilized. It appears that it can be used as a competitive electrode in fuel cells. Nickel foam with its fine structure provides a suitable space for the interaction of light, catalyst, and efficient mass transfer of reactive molecules. In this regard, we investigated the response of a photoanode covered with UiO66-NH 2-TiO 2/NiF on a porous nickel foam as an attractive electrochemical response to remove antibiotics from aqueous solution and simultaneously produce electricity using a one-step hydrothermal synthesis. Photocatalytic fuel cell, a potential candidate for converting chemical energy into electrical energy through a pollution-free method, holds promise in addressing these challenges. The ones listed are all ones I actually have in the collection.Environmental destruction, water crisis, and clean energy are among the very important challenges worldwide based on sustainable development goals. I have just duplicated a photo of one of them to use for all of them, because they really do look exactly the same regardless of what element is inside. 
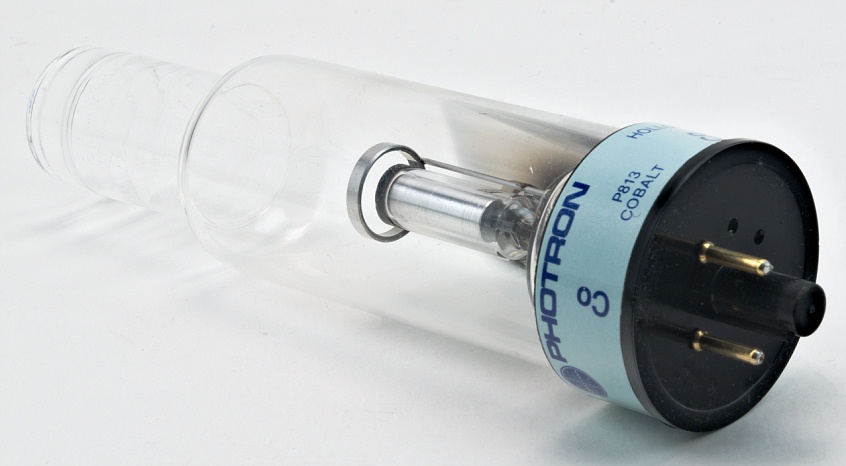
(Truth in photography: These lamps all look alike. I've listed the price of all the lamps as $20, but that's really just a rough average: I paid varying amounts at various eBay auctions for these lamps, which list for a lot more from an instrument supplier. But it does mean that they all look pretty much the same color to the naked eye. In a spectrometer this is no problem because you just use a prism or diffraction grating to separate the light into a spectrum, then block out the neon lines. Neon emits a number of very strong orange-red lines that overwhelm the color of the specific element. Unfortunately, the lamps all use neon as a carrier gas: You generally have to have such a carrier gas present to maintain the electric arc. In theory, each different lamp should produce a different color of light characteristic of its element. The lamp uses an electric arc to stimulate the element it contains to emit its characteristic wavelengths of light: The same electronic transitions are responsible for emission and absorption, so the wavelengths are the same. They are used as light sources for atomic absorption spectrometers, which detect the presence of elements by seeing whether a sample absorbs the very specific wavelengths of light associated with the electronic transitions of the given element. Lamps like this are available for a very wide range of elements: Click the Sample Group link below to get a list of all the elements I have lamps like this for. Hollow cathode lamp, a sample of the element Neon in the Periodic Table H
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
